Physiology of Capnography
Bhavani Shankar Kodali MD
| Fast or Regular Capnogram | A Trend Capnogram |
 |
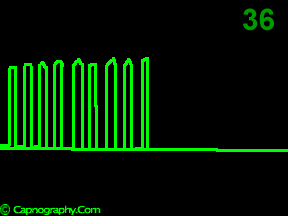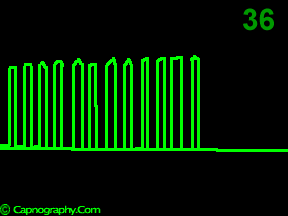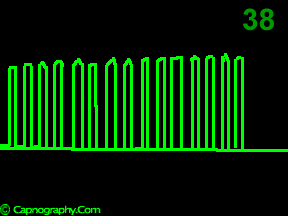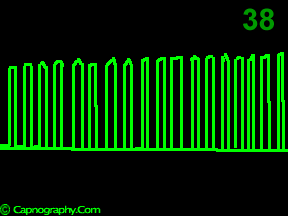 |
A time capnogram may be recorded at two speeds. A high speed capnogram (about 7mm.sec-1) gives detailed information about each breath whereas the overall CO2 changes (trend) can be followed at a slow (about 0.7 mm.sec-1) speed.1
Reference:
1.Bhavani Shankar K, Moseley H, Kumar AY, Delph Y. Capnometry and anaesthesia. Can J Anaesth 1992;39:6:617-32.

 Twitter
Twitter Youtube
Youtube









